Provide your staff with unbiased help while they design, develop, produce, and maintain medical devices. Objective evidence requires documentation. 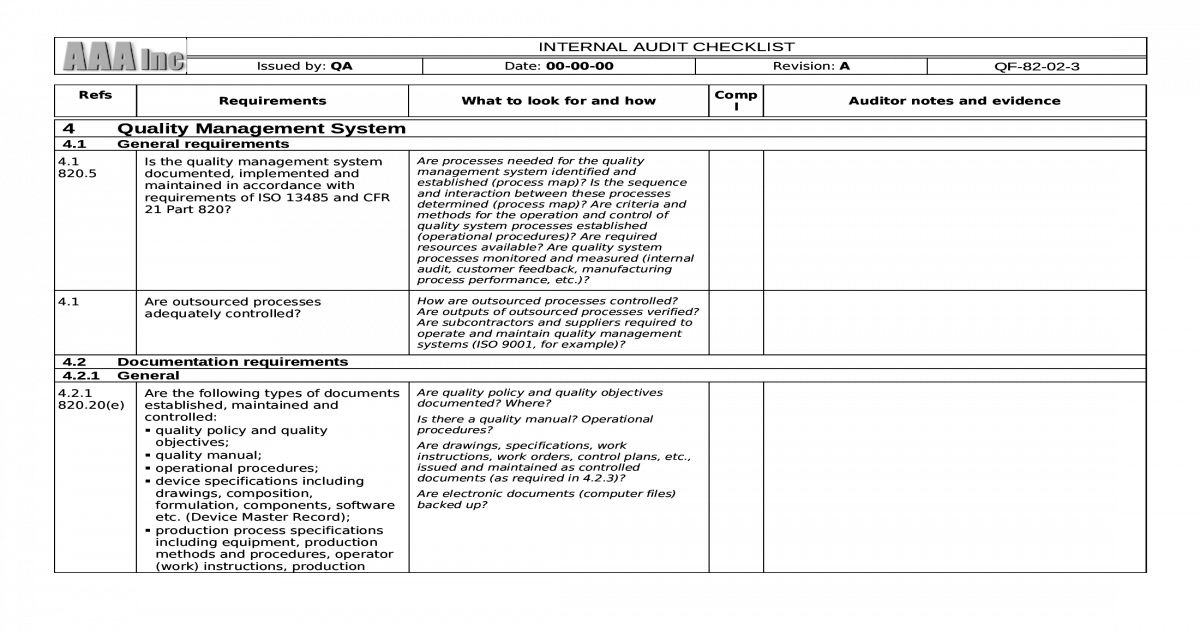
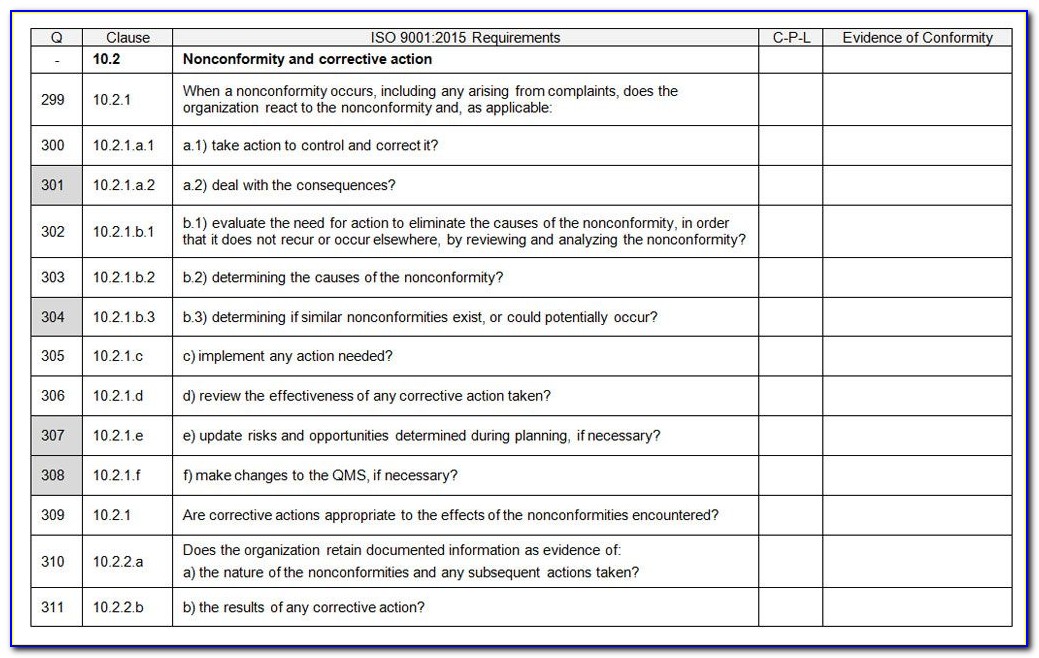
The purpose of documentation should be to specify processes and keep the records necessary to prove they are being followed. So, to assist everyone to understand the ISO 3485 standard better, here are a few of the necessary ISO 13485 documentation requirements:įor every implementation process, documentation of ISO 13485 QMS for medical devices is crucial. However, many businesses begin creating the ISO 13485 Documents before beginning implementation in the organization to gain an idea of what truly needs to be done and how to accomplish it. Because ISO 13485 is an internationally recognized standard of quality and safety for the manufacturing of medical equipment, having it helps firms be perceived as more dependable, trustworthy providers. With the help of the ISO 13485 standard, organizations may create safer and more cost-effective workplaces while also removing any potential legal issues. These specifications are meant to guarantee that medical products and services constantly live up to consumer expectations as well as pertinent legal and regulatory requirements. 
The most recent version of the ISO 13485 standard, becomes applicable in March 2016. The first edition of ISO 13485 was released in 1996, and updates followed in 20. The ISO 13485 standard quality management system (QMS) criteria are a globally accepted set of guidelines for any business engaged in the design, manufacture, distribution, installation, and maintenance of medical devices. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
